1. Intro
The old proverb that a image is worth a thousand phrases certainly does apply to the recognition of phenotypic variations in biomedical research. Bright industry microscopy, by detecting lighting transmitted through slim and clear specimens, provides been broadly used to investigate cell dimension, form, and movement. The recent growth of fluorescent protein, e.g., green fluorescent protein and its derivatives 1, allowed the analysis of the phenotypic changes of subcellular proteins structures, at the.gary the gadget guy., chromosomes and microtubuIes, revolutionizing optical image resolution in biomedical research. Fluorescent protein are destined to particular protein that are usually uniformly located in relevant cellular constructions, e.g., chromosomes, and emit more wavelength lighting, e.g., green light, after exposure to shorter wavelength light, e.g., blue light. Therefore, the spatial morphoIogy and temporal dynamic activities of subcellular proteins buildings can be imaged with á fluorescence microscope - án optical microscope thát can specifically detect emitted fluorescence of a particular wavelength 2. In current image-based research, five-dimensional (5D) image information of hundreds of tissue (mobile populations) can be obtained: spatial (3D), time lapse (1D), and several fluorescent probes (1D).
Tattoo segmentation with unknown number of clusters is transferred to a gure-ground segmentation. We have applied our segmentation algorithm on a tattoo dataset and the results have shown that our tattoo segmentation system is e cient and more » suitable for further tattoo classi cation and retrieval purpose. A standalone simulation toolkit 80 capable of generating 3D images with nuclei forming the villi of human colon tissue employing the model of Voronoi diagram (Fig. 2f) was later proposed. Another study 81 recently extended this principle by generating whole cells, i.e., including the cytoplasm. This method, however, manipulated only 2D image data.
With advances to automatic high-resolution micróscopy, fluorescent labeling, ánd automatic dealing with, image-based research have become well-known in drug and target development. These image-based research are frequently known to as the High Content Evaluation (HCA) 3, which concentrates on removing and examining quantitative phenotypic information instantly from large quantities of mobile pictures with approaches in picture analysis, calculation vision and device understanding 3, 4. Programs of HCA for screening medicines and focuses on are referred to as High Content Screening process (HCS), which focuses on determining compounds or genes that result in desired phenotypic adjustments 5-7. The image data contain rich information content for knowing biological procedures and drug effects, suggest different and heterogeneous behaviours of individual tissues, and provide stronger record power in sketching experimental observations and conclusions, likened to typical microscopy research on a several cells. However, extracting ánd mining the phénotypic changes from the large scale, complex picture data is challenging. It is certainly not feasible to personally analyze these information. Therefore, bioimage informatics approaches were needed to instantly and objectively evaluate large scale picture data, draw out and evaluate the phenotypic adjustments to account the results of medicines and focuses on.
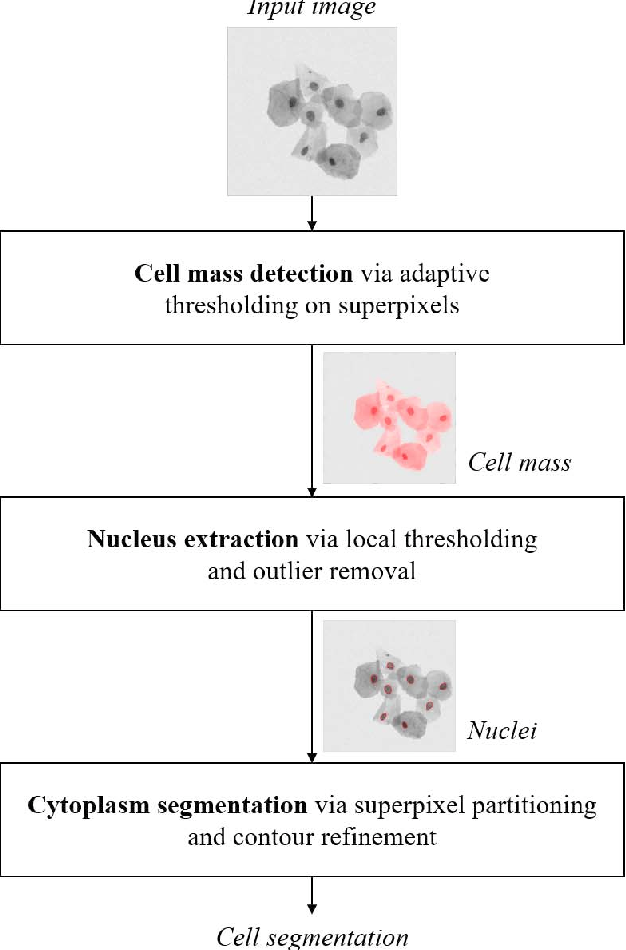
Bioimagé informatics in imagé-based research usually is made up of several analysis segments 3, 8, 9, as shown in Body 1. Each of the evaluation tasks is definitely demanding, and various approaches are often required for the analysis of various forms of images. To assist in image-based screening process research, a number of bioimage informatics software packages have been created and are publicly available 9. This chapter provides an summary of the bioimage informatics approaches in image-based studies for drug and target finding to assist readers, including those without bioimage informatics expertise, know the abilities, techniques, and tools of bioimage informatics and utilize them to move forward their own studies. The rest of this part is structured as follows. Area 2 presents a amount of practical screening applications for finding of potential medications and targets. Section 3 talks about the problems and strategies for quantitative image analysis, e.g., object detection, segmentation, and tracking. Section 4 presents strategies for quantification of segmented objectives, including function removal, phenotype classification, and clustering. Section 5 reviews a quantity of common methods for profiling medication effects centered on the quantitative phenotypic information. Section 6 listings major, openly available software deals of bioimage informatics evaluation, and finally, a brief summary is certainly provided in Area 7.
- bigger image
